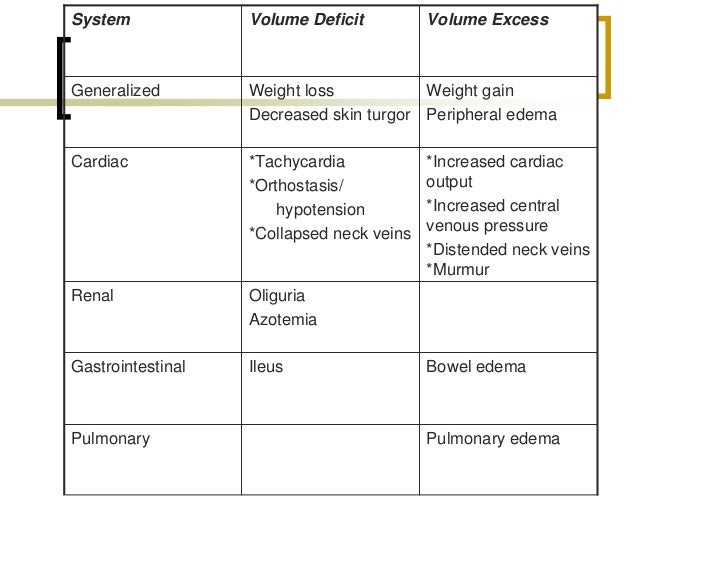
A less commonly appreciated response is transcapillary refill, which involves movement of interstitial fluid into the vascular space to replenish lost intravascular volume ( 16, 17). The defense of ECBV classically involves vasoconstriction, tachycardia, and improved myocardial contractility to maintain circulatory pressure and flow to vital organs. Thus, ECFV and ECBV parallel one another normally, but diverge in many pathologic states for example, edematous states such as congestive heart failure or cirrhosis, often exhibit diminished ECBV with expanded ECFV ( 15). Plasma volume, as a component of blood volume, represents the common link between ECFV and ECBV. Neurohormonal homeostatic mechanisms sense and defend effective circulating blood volume (ECBV), a poorly measurable quality of arterial filling determined primarily by blood volume, cardiac output, and vascular tone ( 15). At the bedside, the measured serum Na + is corrected for its decline related to hyperglycemia so that it largely reflects TBNa + and TBK + relative to TBH 2O: Serum Na + G = Measured serum Na + + (1.7 × ΔGlucose/100 in mg/dL). The clinically relevant range of V D (0.3-0.5) translates into a 1.5-1.9 mEq/L (mean 1.7 mEq/L) fall in serum Na + for every 100 mg/dL rise in serum glucose ( 11- 14).
EPICHROME VS FLUID PLUS
V D is a complex function of insulin activity, glucose distribution time, ECFV, and glucose concentration itself, but under normal steady-state conditions encompasses the mannitol-penetrable ECF plus about 10% of ICF (V D ≅ 0.4) ( 3, 9, 10). A dilutional fall in serum Na + ensues proportional to the change in glucose concentration and V D: ΔSerum Na + G = ΔGlucose × (1-V D) ÷ 2. As glucose accumulates in the extracellular space, effective osmolality rises leading to a shift of body water from ICF to ECF to re-establish equilibrium at a new level of tonicity.

Based on this framework, a modified relationship for serum Na + correcting for hyperglycemia may be derived as follows: Serum Na + G = (TBNa + + TBK + ÷ TBH 2O) − Glucose × (1 - V D) ÷ 2. In the setting of hyperglycemia, the relationship between TBGlucose and serum glucose can be mathematically expressed using a framework in which glucose is added to plasma and allowed to diffuse into a volume of distribution (V D) expressed as a fraction of total body water: Serum glucose = TBGlucose ÷ (V D × TBH 2O). When the content of body solute, levels of anti-diuretic hormone, and extracellular volume are steady-state normal, urine Na + and K + excretion and the electrolyte-free water clearance primarily reflect dietary intake ( 6- 8). TBNa +, TBK +, and TBH 2O are governed by diet and renal excretion, and to a lesser extent by losses from the gut, lungs, and skin. Lest we forget, intracellular K + is an important determinant of steady state serum Na + ( 6, 7), as osmotically active TBK + is 20% more abundant than TBNa +, which explains why the ICF is slightly larger than the ECF ( 4). These bulk solutes obligate water to hydrate one compartment or the other in proportion to available effective osmoles and, at equilibrium, serum Na + roughly reflects net tonicity imposed by effective osmoles across all compartments.
In this thought equation, osmotically-active TBNa + and its anions (not shown) plus glucose bathe the outside of cells and osmotically-active TBK + and its anions are inside cells. If the cell volume effects of tonicity cannot be quantitated directly and serum osmolality is an unreliable indicator ( 7), then serum Na + becomes a useful surrogate marker of tonicity, and we can construct a thought equation to understand this surrogacy: Serum Na + = Tonicity = Effective Osmols ÷ TBH 2O = (TBNa + + TBGlucose + TBK +) ÷ TBH 2O. Ineffective osmoles, such as urea and alcohol, cross cell membranes and do not influence transmembrane water flow or change cell volume ( 5, 6). Only effective osmoles trapped on either side of the cell membrane change cell volume they obligate the hydration of their respective body space through transmembrane water flow until effective osmolality equalizes across all fluid compartments to establish tonicity. Tonicity is different than serum osmolality because measurement of the latter reflects the totality of effective and ineffective osmoles in a liter of body fluid. Tonicity is a descriptive physiologic term that refers to the volume behavior of cells in a solution cell volume tends to expand as body fluids become hypotonic or shrink as surrounding fluids become hypertonic.

Why do we ever measure a serum Na + clinically? Its usefulness lies only as a surrogate marker of tonicity.
